|
3/15/2024 0 Comments Examples of bcs class 1 drugs 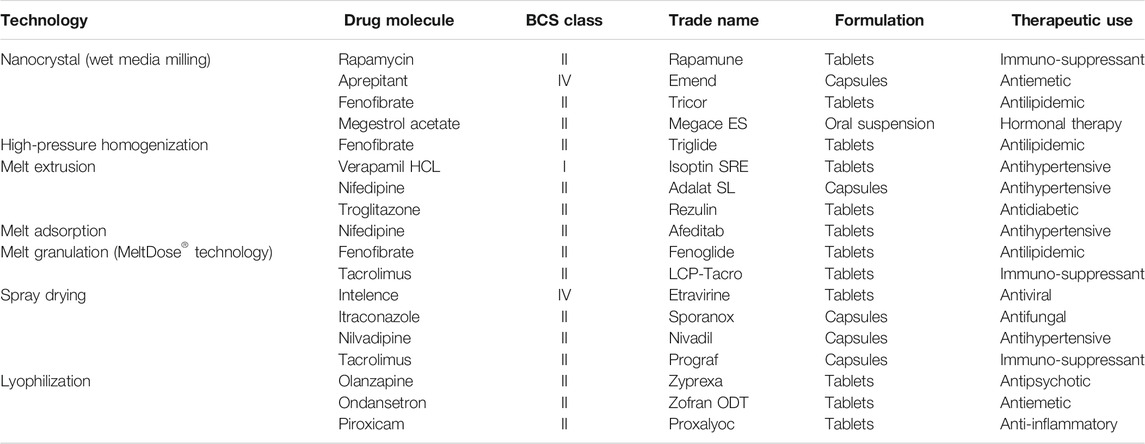
Since the reference product would have been approved on the basis of clinical trials where the safety and efficacy of that product was established, the need to redo expensive and time consuming clinical trials to assess the safety and efficacy is considered unnecessary and instead, a surrogate measure whereby the concentration of drug in serum/plasma of human subjects is compared between the test and reference product is considered more appropriate. The assessment of the safety and efficacy of multisource (generic) products involves a comparative study between the generic (test) products to be registered versus the innovator (reference) where their respective bioavailabilities are compared. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
